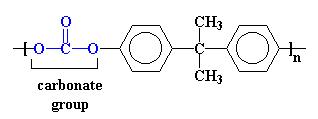
Made by reacting bisphenol and phosgene, or by reacting a polyphenol with methylene chloride and phosgene. The monomer may be OC6H4C(CH3)2C6H4OC:O. The molecular structure is in double-linked zigzag chains that give high rigidity. The resin is thermoplastic. It is crystalline with rhombic crystals.
Polycarbonate is a linear, low-crystalline, transparent, high-molecular-weight plastic. It is generally considered to be the toughest of all plastic. In thin sections, up to about 3/16 in (0.478 cm), its impact strength is as high as 16 ft • lb (24 kg • m). In addition, polycarbonate is one of the hardest plastics. It also had good strength and rigidity, and, because of its high modulus of elasticity, is resistance to creep. These properties, along with its excellent electrical resistivity, are maintained over a temperature range of about —275 to 250°F (—170 to 121°C). It has negligible moisture absorption, but it also has poor solvent resistance, and, in a stressed condition, will craze or crack when exposed to some chemicals. It is generally unaffected by greases, oils, and acids. Polycarbonate plastics are easily processed by extrusion, by injection, blow, and rotational molding, and by vacuum forming.
They have very low and uniform mold shrinkage. With a white light transmission of almost 90% and high impact resistance, they are good glazing materials. They have more than 30 times the impact resistance of safety glass. Other typical applications are safety shields and lenses. Besides glazing, polycarbonate’s high impact strength makes it useful for air-conditioner housings, filter bowls, portable tool housings, marine propellers, and housings for small appliances and food-dispensing machines.
Applications:
· Insulating bolts and nuts
· Valves keyboard cases
· Electric tool housings
· Medical devices
· Cocks
· Cellular phone housings